First Class Info About How To Draw Electron Diagrams

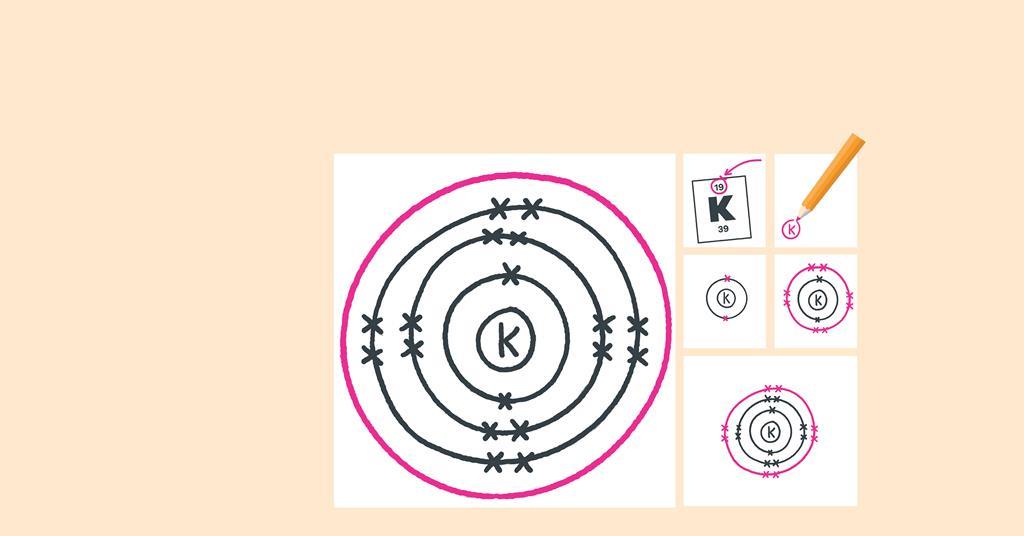
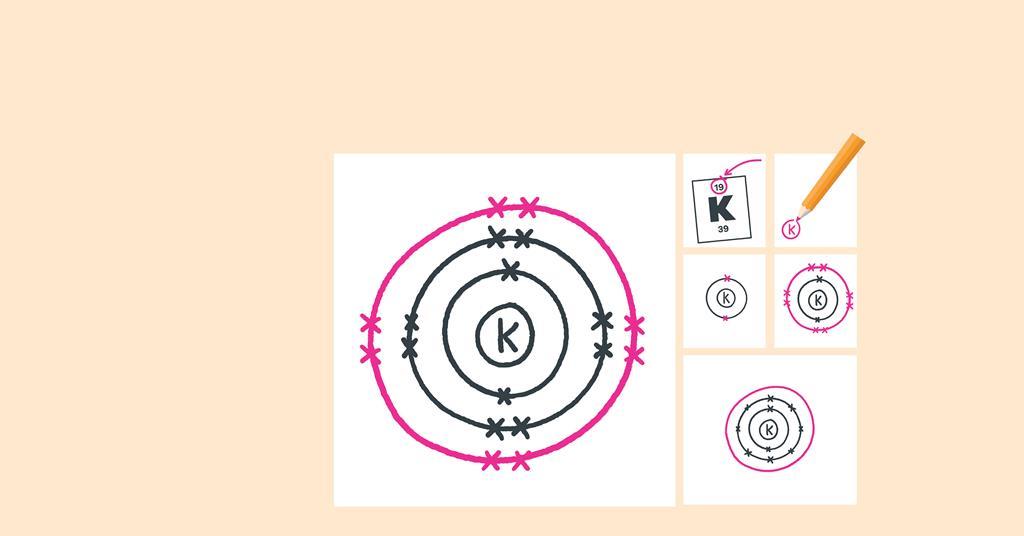
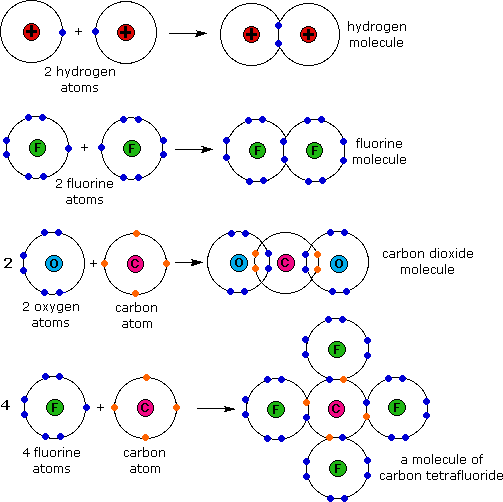
Web electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element’s symbol.
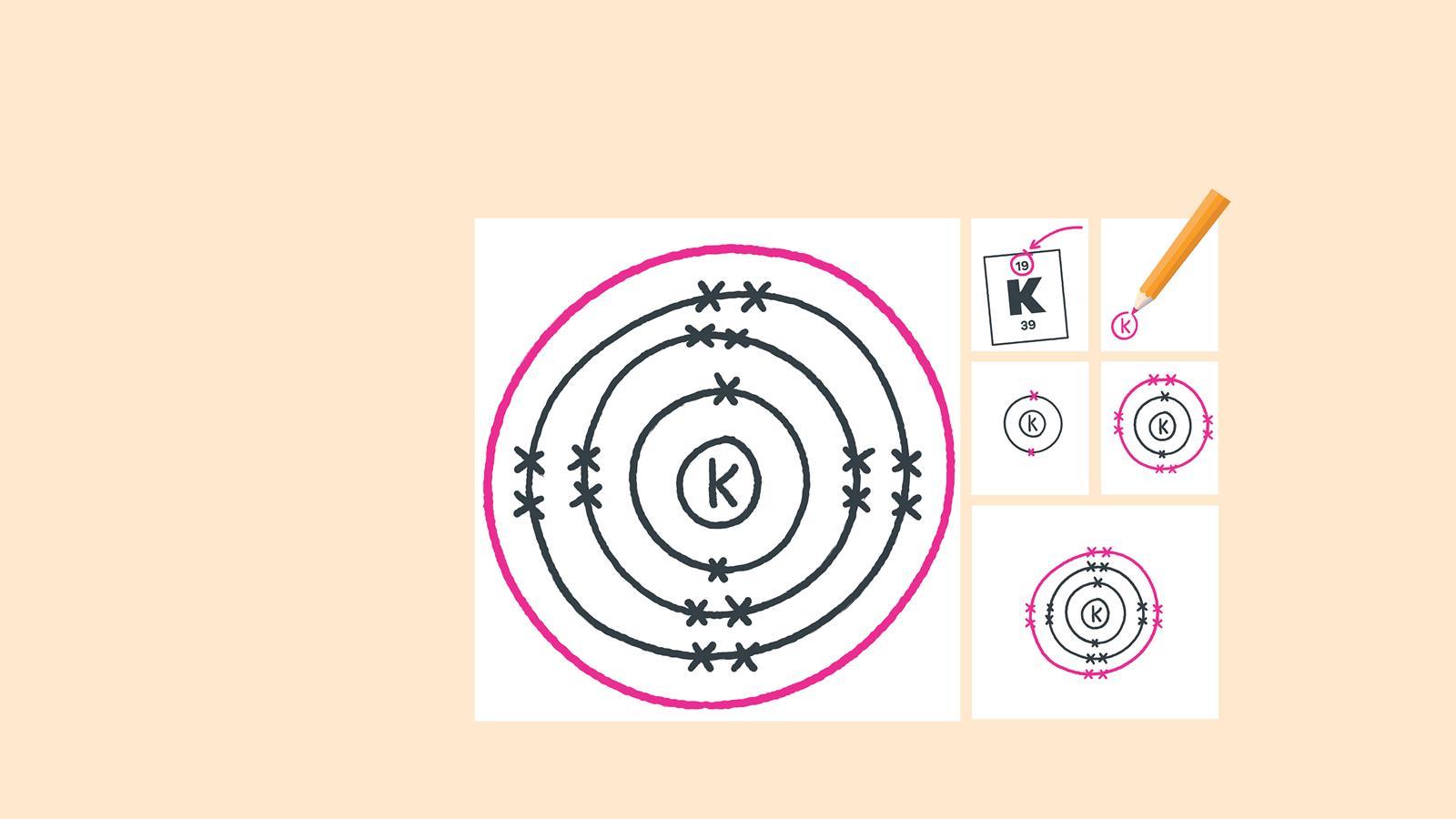
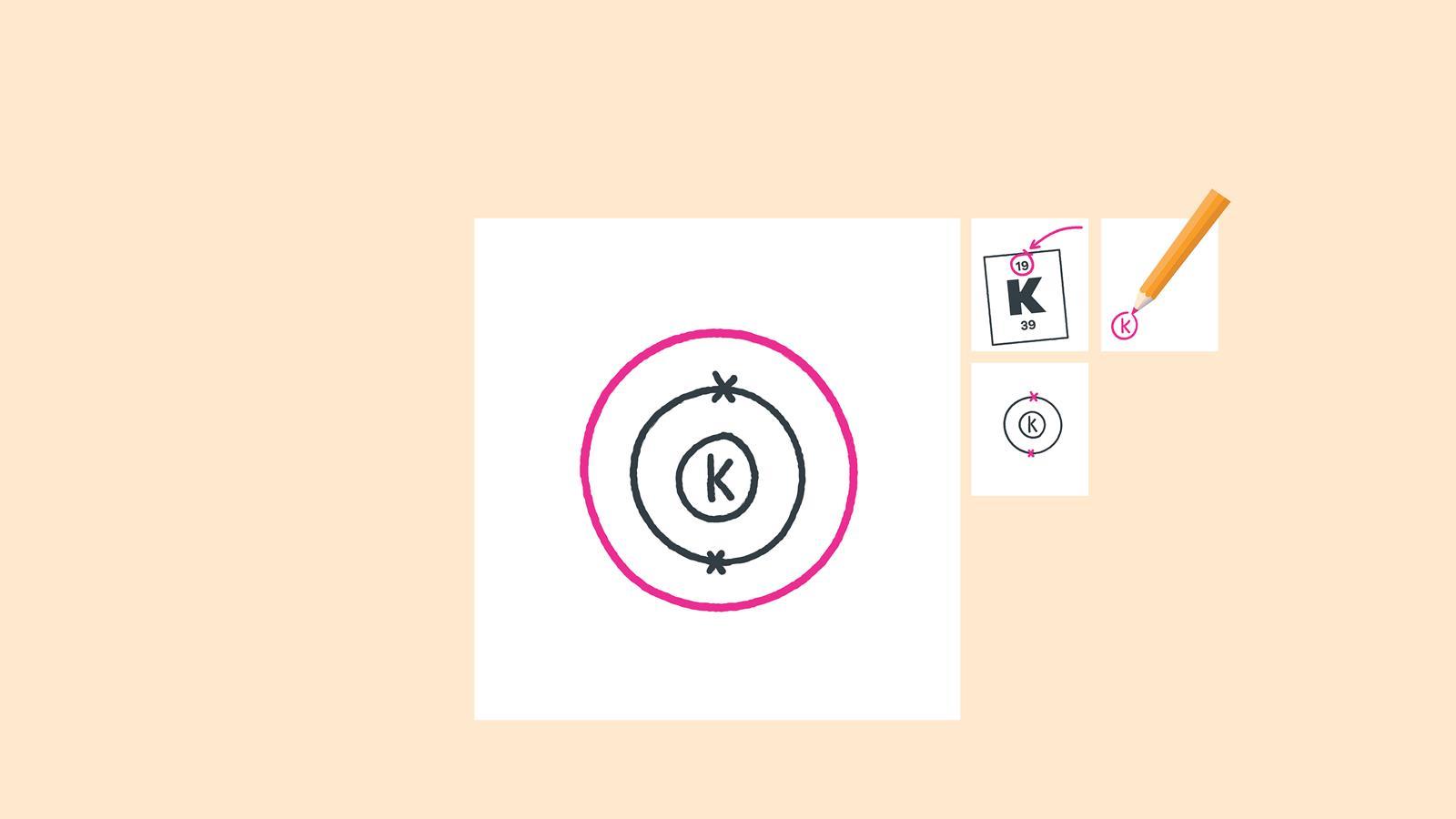
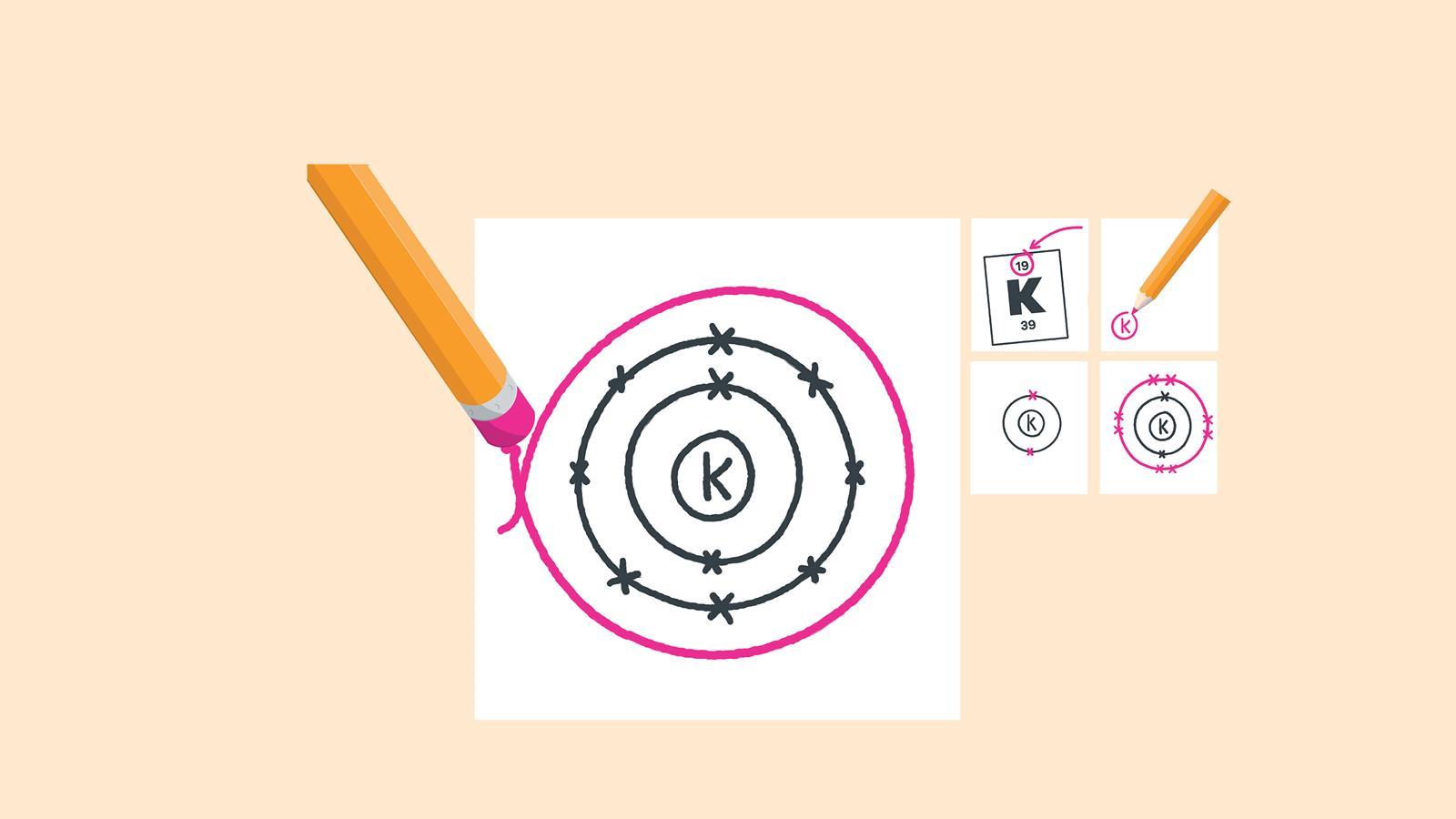
How to draw electron diagrams. Web drawing lewis dot symbols or electron dot diagrams is an important skill in understanding molecular geometry and ionic crystals. It’s a good idea to draw the electrons in pairs but remember. Draw the nucleus of an atom.
Web see the following examples for how to draw lewis dot structures for common atoms involved in covalent bonding. Draw the outer shell of each atom. It explains how to write the orbital diagram n.
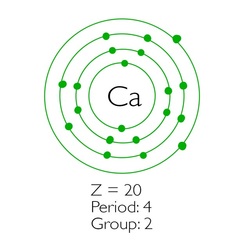
Web to draw the bohr model of an atom, follow these basic steps. Put the least electronegative atom in the center. Web the first number is the principal quantum number (n) and the letter represents the value of l (angular momentum quantum number;
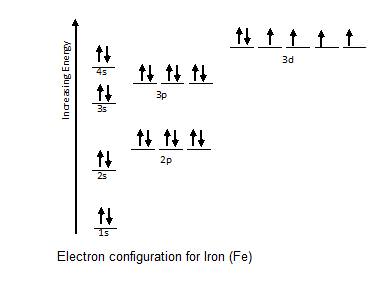
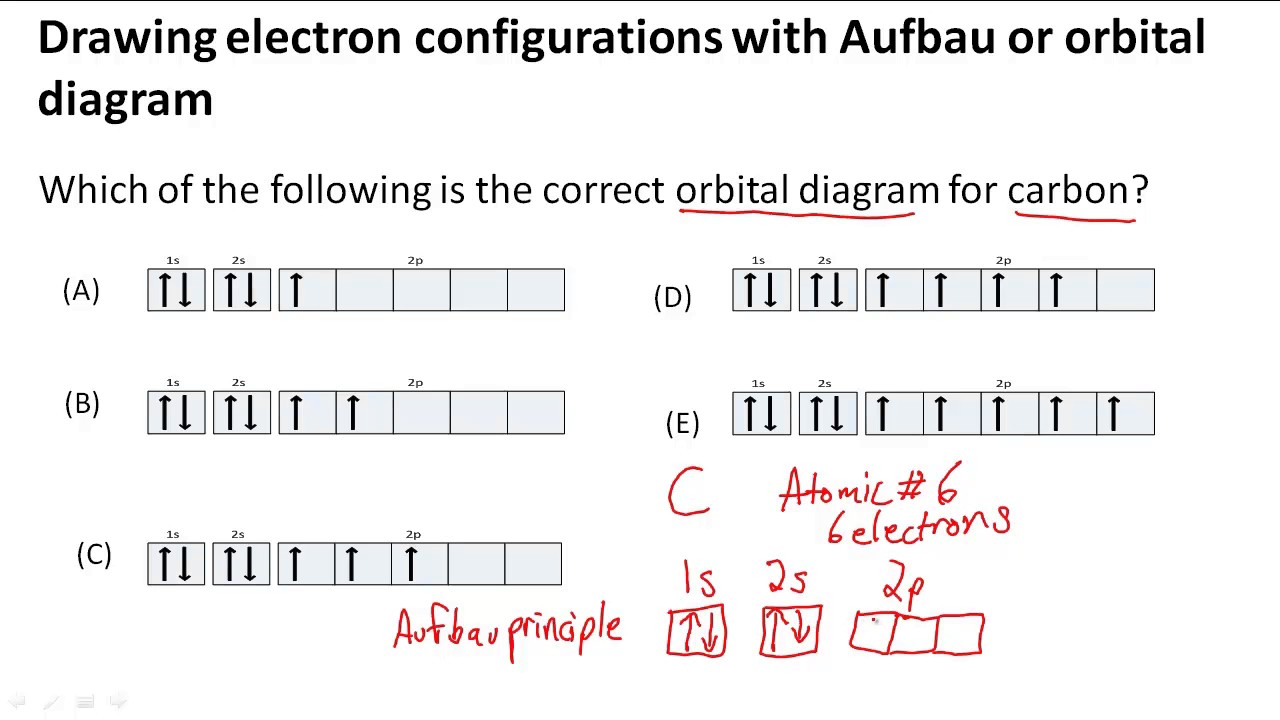
When drawing an element's electron configuration, arrows are used to represent electrons, and lines or boxes are used to represent the orbitals. To write electron configurations and draw orbital box diagrams, there are three rules that must be applied. Web this chemistry video tutorial provides a basic introduction into orbital diagrams and electron configuration.
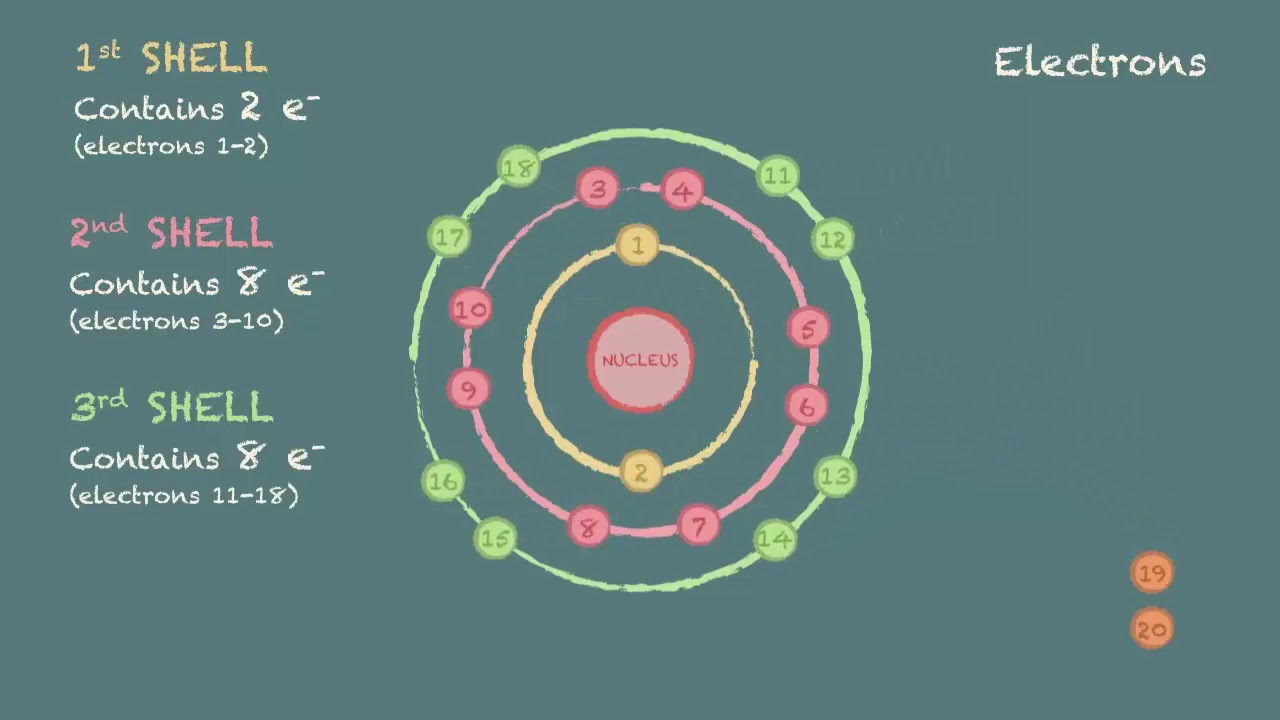
A beryllium atom, with two valence. 1 = s, 2 = p, 3 = d and 4 =. Web sketch out the electron configuration diagrams for each of the atoms.
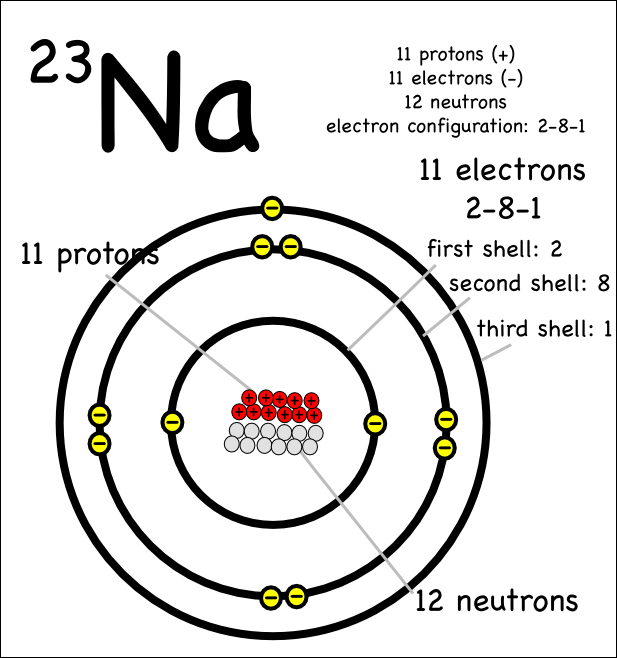
Web write the symbol of the atom you are drawing the electron dot diagram for in the middle of your paper. Web for drawing the orbital diagram or orbital notation, first, find the number of electrons in an atom then write its electron configuration to determine which orbital should be filled. Find the number of protons, electrons, and neutrons of an atom.
This symbol represents the nucleus of the atom and each of the. Determine the total number of valence electrons in a molecule 2. Web bonded pairs of electrons are shown as lines, signifying a covalent bond.
Find the total valence electrons for the molecule. It also helps with understan. Hydrogen (h) always goes outside.
A single line represents a single covalent bond with just one bonded pair of electrons, whilst two lines. Web how to draw orbital diagrams.